A method to calculate new valence electronegativity and the ionicity of chemical bond of the transition metal elements is also presented here.
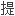

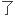 计算过渡元
计算过渡元
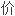
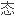 共
共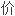 半
半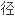 的公式;
的公式;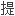

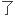 计算过渡元
计算过渡元 新
新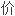
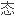 电负性和化学键的离子性的方法。
电负性和化学键的离子性的方法。
声明:以上例句、词性分类均由互联网资源自动生成,部分未经过人工审核,其表达内容亦不代表本软件的观点;若发现问题,欢迎向我们指正。
 院:生物化
院:生物化
 院:生物化
院:生物化
 Crash Course
Crash Course  院:生物化
院:生物化
 院:生物化
院:生物化
 院:生物化
院:生物化
 院:生物化
院:生物化
 院:生物化
院:生物化
 院:生物化
院:生物化
 院:生物化
院:生物化
 Crash Course
Crash Course  院:生物化
院:生物化
 Crash Course
Crash Course  院:生物化
院:生物化
 院:生物化
院:生物化